Regenesis (6 page)
Authors: George M. Church
Figure 1.3
The mirror forms of tartaric acid at the atomic level on the left and the corresponding crystals of the salts: for each pair the natural one is on the left. The handedness at the atomic (nanometer) scale building blocks is amplified up to the human (millimeter) scale, a key requirement at the time of Pasteur, since the ability to manipulate molecules was still quite primitive. Just as the arrangement of the atoms of the two molecules can only be superimposed by use of mirrors, not by mere rotations, the same is true of the arrangement of the faces of the two crystals.
The future of chirality is bright with promise and includes the synthesis of mirror cells that will give us access to valuable chemicals and materials, as well as cells that resist most biological degradation. Heat can often reverse the handedness of a given molecule, and a class of enzymes called racemases can do the same. The most obvious way of creating a mirror cell would be to synthesize unnatural mirror versions of all living molecules and then assemble the parts in the correct spatial arrangements into a full cell and hope that it replicates.
The enormity of such a task will become more evident in
Chapter 2
, where I describe modern cells and minimal versions of cells. I show a possible shortcutâthat by leveraging a slightly sloppier version of current life we could avoid the precise manual synthesis and assembly of billions of chemical bonds. But for now we want to consider the implications, regardless of route. The complete synthesis of a mirror life form from the atoms
up would be the next, and perhaps final, step in the overthrow of vitalism. The shortcut version would pose less of a threat to vitalism but would be just as significant in terms of its potential future applications.
Creating a mirror world might give us a fresh lease on life, one free of disease and unwanted agricultural pest species, but subject to unintended consequences such as turning too much carbon dioxide into RDOM or encouraging the proliferation of enzymes that could attack our wonderful new mirror lifeâenzymes that are currently rare but in a sense are waiting for a justification for their duplication, diversification, and optimization. Unlike antibiotics, of which there are thousands of natural and totally synthetic examples to draw upon, there are only two hands, and we've already “used up” one of them.
Another profound implication is that we are doubling the number of chiral chemicals in our bag of tricks. Because many chemicals have more than one chiral atom, the number of new compounds might be exponentialâas high as 2
N
where there are N atoms with the property of mirror asymmetry. This could be especially apparent in polymersâthe topic of the next section.
Having journeyed from inorganic to organic and having considered the handedness of simple monomers, we now take a look at polymers, the next big idea in the story of the past and future of life. The key substances in most modern biological structures and catalysts are proteins, which are polymers made up of the twenty or so amino acids. The other key classes of polymers are the polynucleotides DNA and RNA, each of which is composed of four nucleotides (adenine, cytosine, guanine, and thymine in DNA, with uracil replacing thymine in RNA). Like proteins, DNA and RNA have handedness; Tom Schneider (a molecular-machine researcher at the US National Cancer Institute) maintains a web page (
http://www.fred.net/tds/leftdna
) dedicated to seven hundred humorous examples of
wrong-handed DNA helix art in the popular press and in company brochures. (We need to create mirror DNA to make these folks seem more prescient and less foolish!) Like polypeptides, polynucleotides are capable of structural scaffolding and catalysis but have the additional feature of making replication possible (indeed obvious and compelling).
The story of DNA (and RNA) replication is beautifully simple. The core idea is that of complementary surfaces. Just as the pairs of hands and molds in the sculpture shown in
Figure 1.1
are uniquely complementary, so too are the base pairs of RNA: U (uracil) bonds with A (adenine), and C (cytosine) with G (guanine),
Figure 1.4
. The idea that a DNA duplex explains DNA duplication, and these two base pairs in particular, won Jim Watson and Francis Crick the Nobel Prize. By contrast, the other eight possible pairings (AA, AC, AG, CC, CU, GG, GU, and UU) are much weakerâenergetically close to no pairing at all. In the case of the strong pairings, the two surfaces fit together, so to speak, whereas in the case of the weak pairings, there is a mismatch between the respective molecular shapes.
The stability of two strands of RNA binding to each other depends essentially on their length. Given a long polymer of nucleotides, we can assemble matching monomers, or short complementary polymers, onto it by base pairing. The stability of the resulting double helices gives the second strand of RNA a chance to polymerize or ligate (join) the short bits into a new long polymer (probably catalyzed by molecules floating about). This new polymer is not identical to the original but complementary, but the complement of the complement is the original.
Earlier I asked how we can get from atoms to replicable structures from scratch, meaning from atoms or tiny clusters of atoms without assistance from living templates. We can now see a progression of events from the beginning of this chapter as atomic nuclei randomly join to become atoms, atoms join to become molecules, and monomeric molecules randomly join up to become polymers. The chemical bonding ratios up to this point seem predetermined by physical selection rules acting on large sets of atoms. The matter that we see in the cosmos requires for its existence only a one part per billion excess of matter over antimatter in the early universe.
Had there been exactly equal amounts, they would have annihilated one another. There is no consensus yet on the explanation of this asymmetry. Similarly, if there had been equal amounts of left- and right-handed molecules, life might not exist in the universeâat least not life as we know it. In any case, once we get replication, then we can expect to see, more and more frequently, small random events that can grow exponentially into interesting structures before any competing chemistry can take hold.
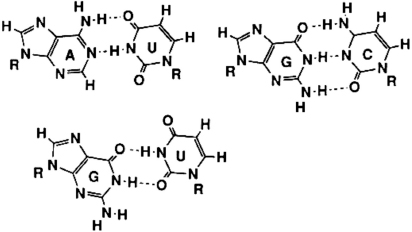
Figure 1.4
Complementary shapes of RNA base pairs. Shown at the top are the two dominant base pairs (AU and GC); just below them is an example (GU) of the other eight possible (very weak) base pairs. What seems like a subtle difference in geometry between the AU pair and the GU mispair makes for a huge difference in the context of a stack of these flat base pairs in the double helix. (The R represents the ribose sugar to which all four bases bind with very similar geometry.) The dotted lines are bonds mediated by hydrogen that are about one hundred times weaker than the covalent bonds (solid lines) in their optimal configuration.
Had there been exactly equal amounts, they would have annihilated one another. There is no consensus yet on the explanation of this asymmetry. Similarly, if there had been equal amounts of left- and right-handed molecules, life might not exist in the universeâat least not life as we know it. In any case, once we get replication, then we can expect to see, more and more frequently, small random events that can grow exponentially into interesting structures before any competing chemistry can take hold.
Evolution happens not only in nature but also in the laboratory, where the key processes of mutation and selection operate on inanimate molecules and structures made up of them. Even creationists can see how small changes, when made repeatedly over long stretches of time, can add up to enormous effects that confer substantial selective advantages on a given organism. What is more remarkable is how new kinds of functionality and shape can emerge out of totally random collections of RNA rather than
as mere variations on something already optimized and working. This process of emergence has major implications for how quickly new genes and genomes could have arisen in the past, as well as for the design of medical and industrial materials in the near future. Totally random libraries of RNA can be subjected to powerful selection pressures that favor rare molecules capable of valuable binding or catalysis functions. We can generate an incredible number of different RNA structures in a volume equivalent to that of a small cell. If any of these
RNAs
has any activity for preferentially cutting and/or joining, then the whole set of RNA sequences could churn and self-modify until stable self-replicating molecules arise and persist.
So, the answer to the question posed earlierâCan a synthetic chemical copy itself and evolve without help from living systems?âis a resounding yes. Here is an example of such evolution in the lab. A molecule of theophylline (which is used as a drug to treat asthma and other lung diseases) can form part of a fifty-five-nucleotide-long stretch of RNA that can have two different morphologies and two different functional states depending on the concentration of theophylline. It is easy to imagine that this molecule could start with either state as its “only” shape and function and could change to the bi-stable shape with as little as the mutation of a single nucleotide. Then after some other molecule adapts to the bi-stable state, another point mutation locks it into one state or the other, permanently.
The moral of the story is that shape and function can be altered radically with just a few changes that nevertheless yield a selective advantage at each separate stage. This capacity will be very handy in the future of lab-evolved designs.
We have been focusing on inorganic and organic chemistry. In colloquial usage the term “organic” is attended by a certain halo effect that, upon analysis, it doesn't deserve. When we buy organic produce, we are supporting the idea of feeding crops the essential elements nitrogen and phosphorus that are derived only from animal excrement rather than from
conventional mineral fertilizers like ammonium phosphate as churned out by the chemical industry. Does this sound like a latter-day vestige of vitalism? These organic fertilizers obviously bear a public health risk in the form of fecal pathogens such as
E. coli
0157:H7,
Cryptosporidium
, and
Gi-ardia
. Both methods of fertilization, if used to excess or done poorly, carry a risk of run-off into streams and ponds resulting in fish kills.
Another inorganic/organic dualism can be seen at the interface between life and machines. I/O means not only the intimate dance of inorganic/organic, but also input/output. Today scientists are recapitulating what we might call the first inorganic/organic transition that occurred eons ago. We take simple molecules and form them into linear polymers that are the building blocks of both natural and synthetic structures. We increasingly want to see input/output between inorganic electronics and organic DNA. On the input side of I/O, megapixel CCD (charge-coupled device) and CMOS (complementary metal oxide semiconductor) electronic cameras can be used to record spatially patterned light, such as bioluminescence or fluorescence, to inorganic (i.e., silicon-based) computers. This would allow us to read genomes speedily, whether for diagnostic testing or environmental monitoring. Coupling these inorganic/organic, input/output features together permits us to design, synthesize, and assess the quality of large collections of DNA and anything that they encode.
Back in the early stone age of DNA engineering (circa 1967â1990) we made DNA in solution and had to purify very short intermediate products. The low yields for each step, multiplied by the short lengths per step, made DNA synthesis a challenging, tedious enterprise. Nowadays we can literally “print” arrays of DNA by machine. This is a really big deal. To see why, let's explore analogies with other types of printing.
Today we use spatially patterned light and optics or ink-jet printers to print photographs on paper, which are two-dimensional artifacts. But it is possible for those same ink-jet printers to “print” (i.e., to construct, layer by layer) three-dimensional objects. Ink-jet systems can hold many colors and activate many jets in parallel. If the ink consists of colored minerals or glue, then we can deposit (or “print”) one layer on top of a second layer
(typically 0.1 mm per layer), and then repeat this process successively to create three-dimensional rapid prototypes of artifacts in plastic or plaster.
We can use similar approaches of spatially patterned light or ink jets to build up long chains of DNA called oligonucleotides, or “oligos” (from the Greek
oligos
, for small), up to 300 nucleotides in length. Typically each layer is one nucleotide (= 0.4 nm) thick, with the four ink-jet “colors” (A, C, G, and T) used per layer. By this method we can make millions of different patches of DNA on a 3- by 1-inch glass slide or portion of a larger silicon wafer.