In Search of Memory: The Emergence of a New Science of Mind (25 page)
Read In Search of Memory: The Emergence of a New Science of Mind Online
Authors: Eric R. Kandel
Tags: #Psychology, #Cognitive Psychology & Cognition, #Cognitive Psychology
The finding that neurons are unique and that the same cell appears in the same location in every member of the species led to new questions: Are the synaptic connections between these unique neurons also invariant? Does a given cell always signal exactly the same target cell and not others?
To my surprise, I found that I could readily map the synaptic connections between cells. By inserting a microelectrode into a target cell and stimulating action potentials in other cells of the ganglion, one cell at a time, I could identify many of the presynaptic cells that communicate with the target cell. Thus it proved possible for the first time in any animal to map the working synaptic connections between individual cells, which I could use as a method for working out the neural circuit controlling a behavior.
I found the same specificity of connections between individual neurons that Santiago Ramón y Cajal had found between populations of neurons. What’s more, just as neurons and their synaptic connections are exact and invariant, so, too, the function of those connections is invariant. This extraordinary invariance would make it easier for me to realize my long-term goal of “trapping” learning in a simple set of neural connections in order to look at how learning gives rise to memory at the cellular level.
By 1969 Kupfermann and I had succeeded in identifying most of the nerve cells that make up the gill-withdrawal reflex. To do this we briefly anesthetized the animal so that we could make a small incision in its neck, and then gently lifted the abdominal ganglion and its attached nerves out through the opening and put them on an illuminated stage. We inserted into various neurons the double-barreled microelectrodes we used for recording and stimulating a cell. Opening up the living animal in this way allowed us to keep its nervous system and all its normal connections intact and thus to observe all of the organs controlled by the abdominal ganglion at the same time. We first set about searching for the motor neurons that control the gill-withdrawal reflex—that is, the motor cells whose axons lead outward from the central nervous system to the gill. We did this by stimulating one cell at a time with the microelectrode and watching to see if that stimulus produced a movement of the gill.
One afternoon in the fall of 1968, working alone, I stimulated a cell and was astonished to see that it produced a powerful contraction of the gill (figure 13–5). For the first time I had identified a motor neuron in
Aplysia
that controlled a specific behavior! I could hardly wait to show Irving. We both were amazed to see the powerful behavioral consequences of stimulating a single cell and knew it boded well for identifying other motor cells. Indeed, within a few months Irving had found five other motor cells. We suspected that those six cells accounted for the motor component of the gill-withdrawal reflex because if we prevented the cells from firing, no reflex response occurred.
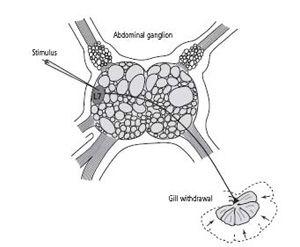
13–5 Discovering a motor neuron that produces a specific behavior in
Aplysia
. Once the individual nerve cells in
Aplysia
’s abdominal ganglion had been identified, it became possible to map their connections. For example, stimulating cell L7 (a motor neuron) produces a sudden contraction of the animal’s gill.
In 1969 I was joined by Vincent Castellucci, a delightful and highly cultivated Canadian scientist with an extensive background in biology who regularly trounced me in tennis, and by Jack Byrne, a technically gifted graduate student with training in electrical engineering who brought the rigor of that discipline to bear on our joint work. Together, the three of us identified the sensory neurons of the gill-withdrawal reflex. We then discovered that in addition to their direct connections, the sensory neurons formed indirect synaptic connections with motor neurons through interneurons, a type of intermediary neuron. Those two sets of connections—the direct and indirect—relay information about touch to the motor neurons, which actually produce the withdrawal reflex by means of their connections with gill tissue. Moreover, the same neurons were involved in the gill-withdrawal reflex in every snail we studied, and the same cells always formed the same connections with one another. Thus, the neural architecture of at least one behavior of
Aplysia
was amazingly precise (Figure 13–6). In time, we found the same specificity and invariance in the neural circuitry of other behaviors.
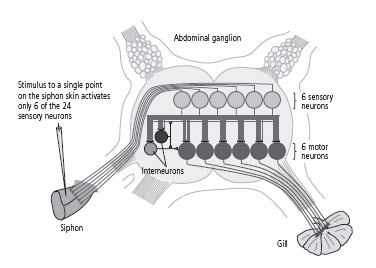
13–6 The neural architecture of
Aplysia’s
gill-withdrawal reflex.
The siphon system has 24 sensory neurons, but a stimulus applied to any one point on the skin activates only 6 of them. The same 6 sensory neurons relay the sensation of touch to the same 6 motor neurons in every snail, producing the gill-withdrawal reflex.
Kupfermann and I ended our 1969 paper in
Science
, “Neuronal Controls of a Behavioral Response Mediated by the Abdominal Ganglion of
Aplysia
,” on an upbeat note:
In view of its advantages for cellular neurophysiological studies, this preparation may prove useful for analyzing the neuronal mechanisms of learning. Initial experiments indicate that the behavioral reflex response can be modified to show simple learning such as sensitization, habituation…. It may also prove possible to study more complex behavioral modifications using either classical or operant conditioning paradigms.
O
nce we had determined that the neural architecture of a behavior is invariant, we were faced with a critical question: How can a behavior that is controlled by a precisely wired neural circuit be changed through experience? One solution had been proposed by Cajal, who suggested that learning could change the strength of the synapses between neurons, thereby strengthening communication between them. Interestingly, Freud’s “Project for a Scientific Psychology” outlines a neural model of mind that includes a similar mechanism of learning. Freud postulated that there are separate sets of neurons for perception and memory. The neural circuits concerned with perception form synaptic connections that are fixed, thus assuring the accuracy of our perceptual world. The neural circuits concerned with memory have synaptic connections that change in strength with learning. This mechanism forms the basis of memory and higher cognitive functioning.
The work of Pavlov and the behaviorists and that of Brenda Milner and the cognitive psychologists had led me to the realization that different forms of learning give rise to different forms of memory. I had therefore reformulated Cajal’s idea and used that new insight as a basis for developing analogs of learning in
Aplysia
. The results of that work had shown that different patterns of stimulation alter the strength of synaptic connections in different ways. But Tauc and I had not examined how an actual behavior is changed and therefore had no evidence that learning really relies on changes in synaptic strength.
Indeed, the very idea that synapses could be strengthened by learning and thus contribute to memory storage was by no means generally accepted. Two decades after Cajal’s proposal, the distinguished Harvard physiologist Alexander Forbes suggested that memory is maintained by dynamic, ongoing changes within a closed loop of self-exciting neurons. To support this idea, Forbes cited a drawing by Rafael Lorente de Nó, a student of Cajal, which showed that neurons connect to each other in closed pathways. The idea was further elaborated by the psychologist D. O. Hebb in his influential 1949 book,
The Organization of Behavior: A Neuropsychological Theory
. Hebb argued that reverberatory circuits are responsible for short-term memory.
Similarly, B. Delisle Burns, a leading student of the biology of the cerebral cortex, challenged the idea that physical changes in synapses can serve as a means of memory storage:
The mechanisms of synaptic facilitation which have been offered as candidates for an explanation of memory…have proven disappointing. Before any of them can be accepted as the cellular changes accompanying conditioned reflex formation, one would have to extend considerably the scale of time on which they have been observed to operate. The persistent failure of synaptic facilitation to explain memory makes one wonder whether neurophysiologists have not been looking for the wrong kind of mechanisms.
Some scholars questioned whether learning could take place at all in fixed neural circuits. For them, learning had to be partially or even totally independent of preestablished neuronal pathways. This view was held by Lashley and by some members of an influential group of early cognitive psychologists, the Gestalt psychologists. A variant of this idea was put forward in 1965 by the neurophysiologist Ross Adey. He began his argument by saying that “no neuron in natural or artificial isolation from other neurons has been shown capable of storing information in the usual notion of memory.” He then went on to argue that the flow of current through the space between neurons may carry information that ranks “at least equivalently with neuronal firing in the transaction of information and even more importantly in its deposition and recall.” For Adey, as for Lashley, learning was completely mysterious.
Having worked out the neural circuitry of the gill-withdrawal reflex and determined that it could be modified by learning, my colleagues and I were in a position to ask which, if any, of these ideas had merit. In the first of three consecutive papers we published in the journal
Science
in 1970, we outlined the research strategy that we had used and that was to guide our thinking for the next three decades:
The analysis of the neural mechanisms of learning and similar behavioral modifications requires an animal whose behavior is modifiable and whose nervous system is accessible for cellular analysis. In this and the subsequent two papers, we have applied a combined behavioral and cellular neurophysiological approach to the marine mollusk
Aplysia
in order to study a behavioral reflex that undergoes habituation and dishabituation (sensitization). We have progressively simplified the neural circuit of this behavior so that the action of individual neurons could be related to the total reflex. As a result, it is now possible to analyze the locus and the mechanisms of these behavioral modifications.
In the ensuing papers we established that memory does not depend on self-exciting loops of neurons. For the three simple forms of learning we studied in
Aplysia
, we found that learning leads to a change in the strength of synaptic connections—and therefore in the effectiveness of communication—between specific cells in the neural circuit that mediates the behavior.
Our data spoke clearly and dramatically. We had delineated the anatomical and functional workings of the gill-withdrawal reflex by recording from individual sensory and motor neurons. We had found that touching the skin activates several sensory neurons that together produce a large signal—a large synaptic potential—in each of the motor neurons, causing them to fire several action potentials. These action potentials in the motor neurons produce a behavior—the withdrawal of the gill. We could see that under normal circumstances, the sensory neurons communicate effectively with the motor neurons, sending them an adequate signal to produce the gill-withdrawal reflex.
We now turned our attention to the synapses between the sensory and motor neurons. We observed that when we produced habituation by touching the skin repeatedly, the amplitude of the gill-withdrawal reflex decreased progressively. This learned change in behavior was paralleled by a progressive weakening of the synaptic connections. Conversely, when we produced sensitization by applying a shock to the animal’s tail or head, the enhanced gill-withdrawal reflex was accompanied by a strengthening of the synaptic connection. We concluded that during habituation an action potential in the sensory neuron gives rise to a weaker synaptic potential in the motor neuron, leading to less effective communication, while during sensitization it gives rise to a stronger synaptic potential in the motor neuron, leading to more effective communication.
In 1980 we carried our reductionist approach one step further and explored what happens at the synapses during classical conditioning. Carew and I were joined in this endeavor by Robert Hawkins, an insightful young psychologist from Stanford University. The son of an academic family, he did not need New York to broaden his horizons: he was already a devotee of classical music and opera. A fine athlete, Hawkins had played on the varsity soccer team at Stanford, and he proceeded to focus his athletic passion on sailing.
We found that in classical conditioning, the neural signals from the innocuous (conditioned) and noxious (unconditioned) stimuli must occur in a precise sequence. That is, when the siphon is touched just before the tail is—thus predicting the shock to the tail—the sensory neurons will fire action potentials just before they receive signals from the tail. The precisely timed firing of action potentials in the sensory neurons, followed by the precisely timed arrival of the signals from the tail shock, leads to much greater strengthening of the synapse between the sensory and motor neurons than when signals from the siphon or the tail occur separately, as they do in sensitization.
These several results on habituation, sensitization, and classical conditioning led us irresistibly to think about how genetic and developmental processes interact with experience to determine the structure of mental activity. Genetic and developmental processes specify the connections among neurons—that is, which neurons form synaptic connections with which other neurons and when. But they do not specify the strength of those connections. Strength—the long-term effectiveness of synaptic connections—is regulated by experience. This view implies that the
potential
for many of an organism’s behaviors is built into the brain and is to that extent under genetic and developmental control; however, a creature’s environment and learning alter the effectiveness of the preexisting pathways, thereby leading to the expression of new patterns of behavior. Our findings in
Aplysia
supported this view: in its simplest forms, learning selects among a large repertoire of preexisting connections and alters the strength of a subset of those connections.
In reviewing our results, I could not help being reminded of the two opposing philosophical views of mind that had dominated Western thought from the seventeenth century onward—empiricism and rationalism. The British empiricist John Locke argued that the mind does not possess innate knowledge but is instead a blank slate that is eventually filled by experience. Everything we know about the world is learned, so the more often we encounter an idea, and the more effectively we associate it with other ideas, the more enduring its impact on our minds. Immanuel Kant, the German rationalist philosopher, argued to the contrary, that we are born with certain built-in templates of knowledge. Those templates, which Kant called
a priori
knowledge, determine how sensory experience is received and interpreted.
In choosing between psychoanalysis and biology as a career, I had decided on biology because psychoanalysis, and its predecessor discipline, philosophy, treated the brain as a black box, an unknown. Neither field could resolve the conflict between the empiricist and rationalist views of mind as long as the resolution required a direct examination of the brain. But examining the brain was just what we had begun to do. In the gill-withdrawal reflex of this simplest of organisms, we saw that both views had merit—in fact, they complemented each other. The anatomy of the neural circuit is a simple example of Kantian
a priori
knowledge, while changes in the strength of particular connections in the neural circuit reflect the influence of experience. Moreover, consistent with Locke’s notion that practice makes perfect, the persistence of such changes underlies memory.
Whereas the study of complex learning had seemed intractable to Lashley and others, the elegant simplicity of the gill-withdrawal reflex in a snail enabled my colleagues and me to address experimentally a number of the philosophical and psychoanalytical questions that had led me to biology in the first place. This I found to be both amazing and humorous.
In the third of our reports in
Science
in 1970, we concluded with these comments:
[T]he data indicate that habituation and dishabituation (sensitization) both involve a change in the functional effectiveness of previously existing excitatory connections. Thus, at least in the simple cases,…[t]he capability for behavioral modification seems to be built directly into the neural architecture of the behavioral reflex.
Finally, these studies strengthen the assumption…that a prerequisite for studying behavioral modification is the analysis of the wiring diagram underlying the behavior. We have, indeed, found that once the wiring diagram of the behavior is known, the analysis of its modification becomes greatly simplified. Thus, although this analysis pertains to only relatively simple and short-term behavioral modifications, a similar approach may perhaps also be applied to more complex as well as longer lasting learning processes.
By sticking with a radically reductionist approach—examining a very simple behavioral reflex and simple forms of learning, delineating cell by cell the neural circuit of the reflex, and then focusing on where change occurs within that circuit—I had reached the long-term goal outlined in my grant application to NIH in 1961. I had “trapped a conditioned response in the smallest possible neural population, the connections made between two cells.”
THUS THE REDUCTIONIST APPROACH LED US TO DISCOVER
several principles of the cell biology of learning and memory. First, we found that the changes in synaptic strength that underlie the learning of a behavior may be great enough to reconfigure a neural network and its information-processing ability. For example, one particular sensory cell in
Aplysia
communicates with eight different motor cells—five that produce movement of the gill and three that cause contraction of the ink gland and thus inking. Before training, activation of this sensory cell excited the five gill-innervating motor cells moderately, causing them to fire action potentials and thereby causing the gill to contract. Activation of this same sensory cell also excited the three ink gland-innervating motor neurons but only very weakly, not enough to produce action potentials or to elicit inking. Thus, before learning, gill withdrawal would take place in response to stimulation of the siphon but inking would not. After sensitization, however, synaptic communication between the sensory cell and all eight motor cells is enhanced, causing the three ink gland-innervating motor neurons to fire action potentials as well. Thus, as a result of learning, when the siphon is stimulated, inking will occur along with more powerful gill withdrawal.
Second, consistent with my reformulation of Cajal’s theory and my earlier work with analogs, we found that a given set of synaptic connections between two neurons can be modified in opposite ways—strengthened or weakened—by different forms of learning. Thus, habituation weakens the synapse, whereas sensitization or classical conditioning strengthens it. These enduring changes in the strength of synaptic connections are the cellular mechanisms underlying learning and short-term memory. Moreover, because the changes occur at several sites in the neural circuitry of the gill-withdrawal reflex, memory is distributed and stored throughout the circuit, not at a single specialized site.